kottke.org posts about chemistry
Bloomberg Businessweek dedicated their entire Sept 2, 2019 issue to the periodic table (it’s 150 years old this year) and the elements it contains. From the introductory essay:
Over the past century and a half, but particularly since World War II, scientists and engineers have learned to treat the periodic table like a banquet table-a bountiful spread from which to pluck what they need. There’s scandium in bicycle frames, tin (stannous fluoride) in toothpaste, tungsten in catheters, and arsenic in some computer chips. We are well past the Stone Age, the Bronze Age, and the Iron Age, and into the Everything Age, because almost every entry on the periodic table is being put to some kind of use in today’s economy (excluding synthetic elements that are costly to make and highly radioactive, such as einsteinium).
Cellphones exemplify the complexification. The first ones in the 1980s “were the size of a shoebox and consisted of 25 to 30 elements,” Larry Meinert, U.S. Geological Survey deputy associate director for energy and minerals, said in 2017. “Today, they fit in your pocket or on your wrist and are made from about 75 different elements, almost three-quarters of the periodic table.” That may include tantalum from Rwanda, potassium from Belarus, silver from Mexico, tin from Myanmar, carbon from India, and germanium from China.
Scrolling down on the main story page will take you on a modern-day tour of the periodic table from the lightest elements (hydrogen, helium, lithium) to the heavier ones (uranium, polonium) to some fake ones (adamantium, unobtanium, feminum).
With amazing super slow-motion footage of a match head starting to burn as a backdrop, this video explains the chemical reactions involved in lighting a match.
When the match is struck, a small amount of the red phosphorus on the striking surface is converted into white phosphorus, which then ignites. The heat from this ignites the potassium chlorate, and the match head bursts into flame. During manufacture, the match stick itself is soaked in ammonium phosphate, which prevents ‘afterglow’ once the flame has gone out, and paraffin, which ensures that it burns easily.
(via gizmodo)
IUPAC, the governing body for the official periodic table of elements, has announced the addition of four new elements to the table: ununtrium, ununpentium, ununseptium, and ununoctium. Those are working names…the teams that discovered each element has been invited to name them.
The proposed names and symbols will be checked by the Inorganic Chemistry Division of IUPAC for consistency, translatability into other languages, possible prior historic use for other cases, etc. New elements can be named after a mythological concept, a mineral, a place or country, a property or a scientist.
Ununoctium is so unstable that its half-life is 0.89 milliseconds and only three or four atoms of the substance have been produced in the past 10 years.
Google Research built an interactive periodic table of the elements where you can see the relative amounts of the elements as found in the human body, in the sea, and, most interestingly, by the number of mentions in books.
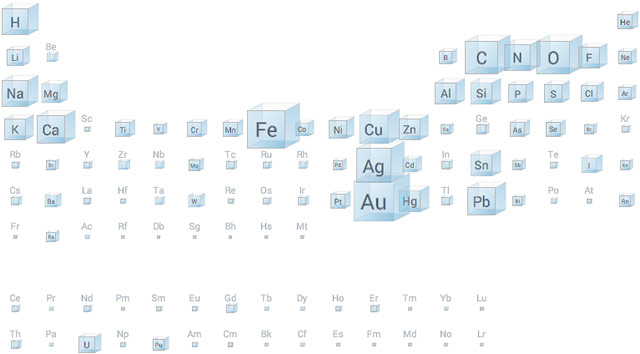
If you’ve ever wondered why the periodic table is shaped the way it is, click on “electrons” under “Shape” and pay attention to the number of electrons in the outer shells in each column of elements. Amazingly, when Dmitri Mendeleev and German chemist Julius Meyer published the first periodic tables in 1869/1870, the elements were organized only by atomic weights and chemical properties; they didn’t know what an electron was and certainly weren’t aware of quantum shells of electrons. (via @djacobs)
Gorgeous videos of chemical reactions (precipitation, bubbling, crystallization, etc.). I think the metal displacement reaction video is my favorite:
(via hn)
The elements located in the upper reaches of the periodic table are notable for their short half-lives, the amount of time during which half the mass of an element will decay into lighter elements (and other stuff). For instance, the longest lived isotope of fermium (#100) has a half-life of just over 100 days. More typical is bohrium (#107)…its half-life is only 61 seconds. The elements with the highest numbers have half-lives measured in milliseconds…the half-life of ununoctium (#118) is only 0.89 milliseconds.
So why do chemists and physicists keep looking for heavier and heavier elements if they are increasingly short-lived (and therefore not that useful)? Because they suspect some heavier elements will be relatively stable. Let’s take a journey to the picturesque island of stability.
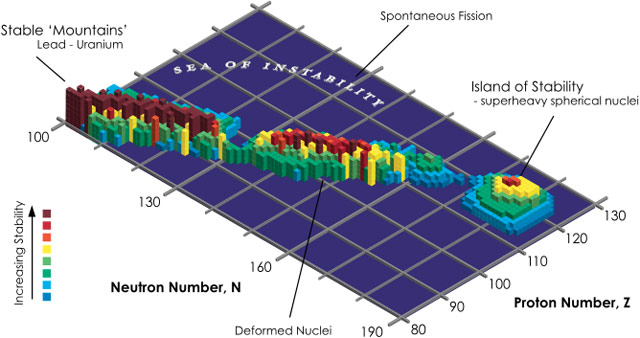
In nuclear physics, the island of stability is a set of as-yet undiscovered heavier isotopes of transuranium elements which are theorized to be much more stable than some of those closer in atomic number to uranium. Specifically, they are expected to have radioactive decay half-lives of minutes or days, with “some optimists” expecting half-lives of millions of years.
Using a modified iPhone and a fluorescing nanoparticle tattoo, researchers at Northeastern University have found a way to monitor chemicals in the blood without drawing blood.
The team begins by injecting a solution containing carefully chosen nanoparticles into the skin. This leaves no visible mark, but the nanoparticles will fluoresce when exposed to a target molecule, such as sodium or glucose. A modified iPhone then tracks changes in the level of fluorescence, which indicates the amount of sodium or glucose present. Clark presented this work at the BioMethods Boston conference at Harvard Medical School last week.
The tattoos were originally designed as a way around the finger-prick bloodletting that is the standard technique for measuring glucose levels in those with diabetes. But Clark says they could be used to track many things besides glucose and sodium, offering a simpler, less painful, and more accurate way for many people to track many important biomarkers.
Sam Kean is blogging the periodic table of elements over at Slate.
Starting today, I’ll be posting on a different element each weekday (the blog will run through early August), starting with the racy history of an element we’ve known about for hundreds of years, antimony, and ending on an element we’ve only just discovered, the provisionally named ununseptium. I’ll be covering many topics-explaining how the table works, relaying stories both funny and tragic, and analyzing current events through the lens of the table and its elements. Above all, I hope to convey the unexpected joys of the most diverse and colorful tool in all of science.
If you like that, Kean has written a whole book on the topic.
Frito-Lay wants to change the shape of the salt they put on their potato chips to increase the surface area exposed to taste buds and therefore decrease the amount of salt needed on each chip.
“Early on in our research, it became apparent that the majority of salt on a snack doesn’t even have time to dissolve in your saliva because you swallow it so rapidly,” explained Mehmood Khan, senior vice president and chief scientific officer and a former Mayo Clinic endocrinologist. A Wall Street Journal story later reported only about 20 percent of the salt on a chip dissolves on the tongue, and the remaining 80 percent is swallowed without contributing to taste.
I’m confused as to why “an understanding of crystal chemistry” is necessary. Why couldn’t they just crush/grind the salt into a fine powder instead? Are the cubic crystals still too big even when crushed?
Electrolyzed water (salt water that has been run through an electrolytic process) is gaining acceptance in the US as a replacement for many cleaning agents.
At the Sheraton Delfina in Santa Monica, some hotel workers are calling it el liquido milagroso — the miracle liquid. That’s as good a name as any for a substance that scientists say is powerful enough to kill anthrax spores without harming people or the environment.
A food science professor says that electrolyzed water is “10 times more effective than bleach in killing bacteria” and it’s safe to drink. (Although maybe it would kill all the bacteria in your stomach?) But beware the phony health claims.
The Periodic Table of Videos is a collection of videos about all the elements. All your favorites are there…Neon, Rubidium, Lead, Plutonium.
I did embarrassingly bad on this Elements of the Periodic Table quiz. I blanked after naming 17 elements in 2 minutes. Oh, and xylophone is not an element! My physics degree should be retroactively unawarded. (via mouser)
How to unboil an egg:
He explains that when an egg is cooked, the protein molecules unroll themselves, link up and enclose the water molecules. In order to ‘uncook’ the egg, you need to detach the protein molecules from each other. By adding a product like sodium borohydride, the egg becomes liquid within three hours. For those who want to try it at home, vitamin C also does the trick.
That’s from an article on Hervé This, a French chemist whose medium is food.
Natalie Angier’s short appreciation of water, which, before you scoff, is a pretty amazing substance despite its ubiquity. “Pulled together by hydrogen bonds, water molecules become mature and stable, able to absorb huge amounts of energy before pulling a radical phase shift and changing from ice to liquid or liquid to gas. As a result, water has surprisingly high boiling and freezing points, and a strikingly generous gap between the two. For a substance with only three atoms, and two of them tiny little hydrogens, Dr. Richmond said, you’d expect water to vaporize into a gas at something like minus 90 degrees Fahrenheit, to freeze a mere 40 degrees below its boiling point, and to show scant inclination to linger in a liquid phase.”
By subjecting ordinary water to extremely high pressure and bombarding it with x-rays, scientists at Los Alamos have formed a new hydrogen-oxygen alloy. “Given high enough pressures, even hydrogen will behave as a metal. All the other heavier elements in hydrogen’s group of the periodic table are metals.”
Philip Stewart has constructed an alternate version of the periodic table of elements in the form of a “chemical galaxy”. “The intention is not to replace the familiar table, but to complement it and at the same time to stimulate the imagination and to evoke wonder at the order underlying the universe.”






Socials & More