The Periodic Table of Endangered Elements
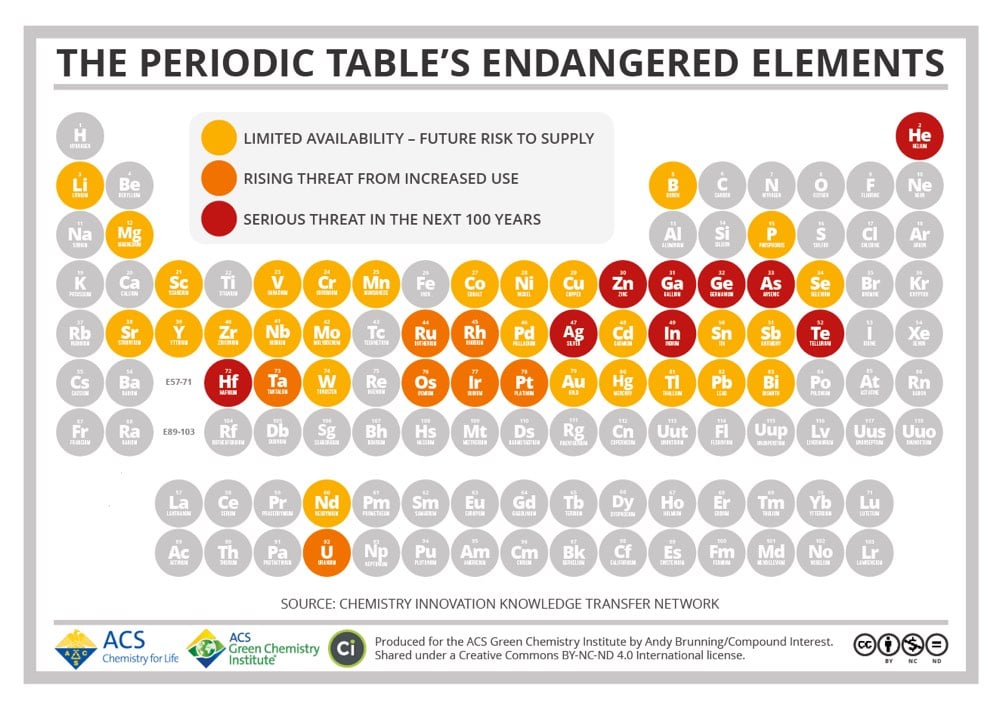
Until recently, humanity has treated the Earth as an infinite resource. As the Earth’s population has exploded over the past century however, we’ve learned in many different ways that that’s untrue. We’ve overfished the ocean, pumped too much carbon into the atmosphere and oceans, driven thousands of species into extinction, and terraformed much of the planet’s land. This periodic table produced by the American Chemical Society shows that there are also 44 chemical elements that will face supply limitations in the coming decades. Among those under a “serious threat in the next 100 years” are silver, helium, zinc, and gallium. Robert Silverberg wrote about The Death of Gallium back in 2008:
Gallium’s atomic number is 31. It’s a blue-white metal first discovered in 1831, and has certain unusual properties, like a very low melting point and an unwillingness to oxidize, that make it useful as a coating for optical mirrors, a liquid seal in strongly heated apparatus, and a substitute for mercury in ultraviolet lamps. It’s also quite important in making the liquid-crystal displays used in flat-screen television sets and computer monitors.
As it happens, we are building a lot of flat-screen TV sets and computer monitors these days. Gallium is thought to make up 0.0015 percent of the Earth’s crust and there are no concentrated supplies of it. We get it by extracting it from zinc or aluminum ore or by smelting the dust of furnace flues. Dr. Reller says that by 2017 or so there’ll be none left to use. Indium, another endangered element-number 49 in the periodic table-is similar to gallium in many ways, has many of the same uses (plus some others-it’s a gasoline additive, for example, and a component of the control rods used in nuclear reactors) and is being consumed much faster than we are finding it. Dr. Reller gives it about another decade. Hafnium, element 72, is in only slightly better shape. There aren’t any hafnium mines around; it lurks hidden in minute quantities in minerals that contain zirconium, from which it is extracted by a complicated process that would take me three or four pages to explain. We use a lot of it in computer chips and, like indium, in the control rods of nuclear reactors, but the problem is that we don’t have a lot of it. Dr. Reller thinks it’ll be gone somewhere around 2017. Even zinc, commonplace old zinc that is alloyed with copper to make brass, and which the United States used for ordinary one-cent coins when copper was in short supply in World War II, has a Reller extinction date of 2037. (How does a novel called The Death of Brass grab you?)
Zinc was never rare. We mine millions of tons a year of it. But the supply is finite and the demand is infinite, and that’s bad news. Even copper, as I noted above, is deemed to be at risk. We humans move to and fro upon the earth, gobbling up everything in sight, and some things aren’t replaceable.
As with many such predictions, the 2017 dates didn’t pan out, but the point that these resources are finite still holds. Eventually, we will run out.




Stay Connected