How the H1N1 vaccine is made
The most striking feature of the H1N1 flu vaccine manufacturing process is the 1,200,000,000 chicken eggs required to make the 3 billion doses of vaccine that may be required worldwide. There are entire chicken farms in the US and around the world dedicated to producing eggs for the purpose of incubating influenza viruses for use in vaccines. No wonder it takes six months from start to finish. But we’ll get to that in a minute.
The most commonly used process for manufacturing an influenza vaccine was developed in the 1940s — one of its co-inventors was Jonas Salk, who would go on to develop the polio vaccine — and has remained basically unchanged since then. The process is coordinated by the World Health Organization and begins with the detection of a new virus (or rather one that differs significantly from those already going around); in this instance, the Pandemic H1N1/09 virus. Once the pandemic strain has been identified and isolated, it is mixed with a standard laboratory virus through a technique called genetic reassortment, the purpose of which is to create a hybrid virus (also called the “reference virus strain”) with the pandemic strain’s surface antigens and the lab strain’s core components (which allows the virus to grow really well in chicken eggs). Then the hybrid is tested to make sure that it grows well, is safe, and produces the proper antigen response. This takes about six to nine weeks.
[Quick definitional pause. Antigen: “An antigen is any substance that causes your immune system to produce antibodies against it. An antigen may be a foreign substance from the environment such as chemicals, bacteria, viruses, or pollen. An antigen may also be formed within the body, as with bacterial toxins or tissue cells.” So, when the H1N1 vaccine gets inside your body, the pandemic strain’s surface antigens will produce antibodies against it.]
At roughly the same time, a parallel effort to produce what are referred to as reference reagents is undertaken. The deliverable here is a standardized kit provided to vaccine manufacturers so that they can test how much virus they are making and how effective it is. This process serves to standardize vaccine doses across manufacturers and takes four months to complete. WHO notes that this part of the process is “often a bottleneck to the overall timeline for manufacturers to generate the vaccine”.
Once the reference virus strain is produced, it is sent to pharmaceutical companies (Novartis, Sanofi Pasteur, etc.) for large-scale production of the vaccine. The companies fine-tune the virus to increase yields and produce seed virus banks that will be used in the bulk production.
And this is where the 1.2 billion chicken eggs come in. A portion of the seed virus is injected into each 9- to 12-day old fertilized egg. The virus incubates in the egg white for two to three days and is then separated from the egg.
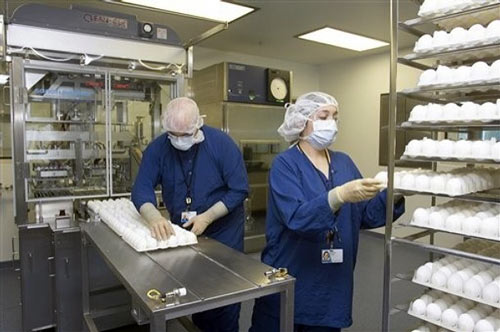
For the shot vaccine, the virus is sterilized so that it won’t make anyone sick. This is the magic part of the vaccine: it’s got the pandemic virus antigens that make your body produce the antibodies to fight the virus but the virus is inactive so it won’t make you ill. For the nasal spray vaccine, the virus is left alive and attenuated to survive only in the nose and not the warmer lungs; it’ll infect you enough to produce antibodies but not enough to make you sick. Either way, the surface antigens are separated out and purified to produce the active ingredient in the vaccine. Each batch of antigen takes about two weeks to produce. With enough laboratory space and chicken eggs, the companies can crank out an infinite amount of purified antigens, but those resources are limited in practice.
[Side note. You may have noticed that the H1N1 vaccine has been difficult to find in some places around the US. The vaccine manufacturers have said that the Pandemic H1N1/09 virus when combined with the standard laboratory virus does not grow as fast in the eggs as they anticipated. The batches of antigens from each egg have been smaller than expected, up to five or even ten times smaller in some cases. Hence the slow rollout of the vaccine.]
The purified antigen is then tested against the aforementioned reference reagents once they are ready. The antigen is diluted to the required concentration and placed into properly labelled vials or syringes. Further testing is performed to make sure the vaccine won’t make anyone ill, to confirm the correct concentration, and for general safety. At this point clinical testing in humans is required in western Europe but not in the United States. Finally, each company’s vaccine has to be approved by the appropriate regulatory body in each country — that’s the FDA in the case of the US — and then the vaccine is distributed to medical facilities around the country.
Sources and more information: WHO, WHO, WHO, WHO, CDC, Time, Washington Post, The Big Picture, Influenza Report, NPR, Wikipedia, Wikipedia, Wikipedia, Wikipedia.
Update: Included in a recent 60 Minutes segment on the H1N1 vaccine is a look at the manufacturing process. (thx, @briandigital)




Socials & More